Carnicom Institute is a non-profit organization working solely for the benefit of humanity. Our goal is to provide the public with beneficial and responsible information through scientific, educational, environmental, and health research for the public welfare. The Institute is currently focused on the important issues of geoengineering and bioengineering.
 |
 |
 |
 |
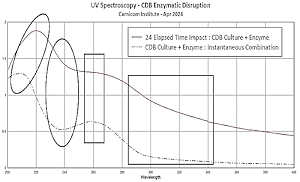
Disruption
Disruption Clifford E Carnicom Apr 23 2024 A measure of success has been achieved at disrupting the early... Read more...more
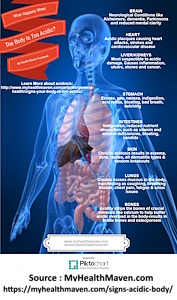
Acids to the Demise
Acids to the Demise Clifford E Carnicom Apr 07 2024 An additional fundamental aspect of the Cross Domain Bacteria... Read more...more
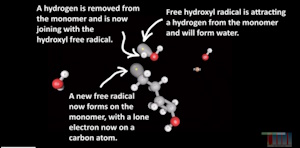
The Polymerization (Clotting) of Blood : A Model
The Polymerization (Clotting) of Blood : A Model Clifford E Carnicom Apr 01 2024 The Polymerization (Clotting) of... Read more...more
Four Mechanisms and a Future for Mitigation (or Termination)
Four Mechanisms and a Future for Mitigation (or Termination) Clifford E Carnicom Mar 14 2024 Carnicom Institute (CI) has... Read more...more
Round 3, Polymer 3: Iron to the Fore
Round 3, Polymer 3: Iron to the Fore Clifford E Carnicom Mar 11 2024 A third polymer form has... Read more...more
Pandora’s Polymer : Synthetic Blood and the Cross Domain Bacteria (CDB)
Pandora’s Polymer : Synthetic Blood and the Cross Domain Bacteria (CDB) Clifford E Carnicom Feb 29 2024 A... Read more...more
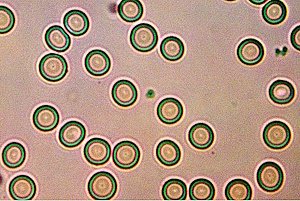
A Cross Domain Bacteria (CDB) Production: (Otherwise Known as Hydrogel)
A Cross Domain Bacteria (CDB) Production: (Otherwise Known as Hydrogel) Clifford E Carnicom Feb 22 2024 CDB Polymer... Read more...more
Blood, Covid & Cross Domain Bacteria (CDB) : Emerging Relationships
Blood, Covid & Cross Domain Bacteria (CDB) : Emerging Relationships Clifford E Carnicom Feb 15 2024 There are some relationships... Read more...more
Bedtime Efforts : Organic Summary II
Bedtime Efforts : Organic Summary II Clifford E Carnicom Feb 02 2024 The following does not qualify as bedtime reading,... Read more...more
A “Filaments” Perspective : 25 Years and Counting…
A “Filaments” Perspective : 25 Years and Counting… Clifford E Carnicom Jan 13 2024 For the past several years, there... Read more...more